Key takeaways
- Radiology has the highest density of FDA-cleared AI devices of any medical specialty — over 700 of the 1,000+ AI devices authorized by FDA as of 2025 are for imaging, per the FDA AI-Enabled Medical Device List.
- Modern radiology AI is built on convolutional neural networks and, increasingly, vision transformers trained on hundreds of thousands of labelled scans.
- Deployed use cases span triage (flag critical findings for fast read), detection (find lesions the radiologist might miss), and quantification (measure tumor size, bone density, cardiac function).
- Despite a decade of “radiologists will be replaced” predictions, radiology job demand has risen; AI works best as a second reader and workflow accelerator.
- Major deployment challenges are workflow integration, payer reimbursement, and validation across diverse patient populations and scanner types.
Why radiology went first
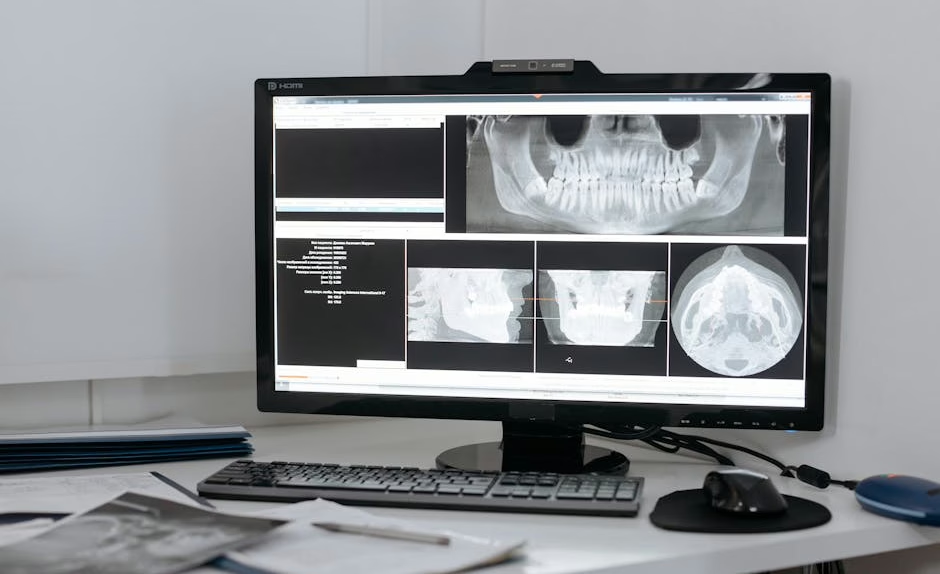
Three things made radiology the natural entry point for medical AI. First, the output of a radiology exam is a digital image — directly consumable by computer-vision models. Second, the task is well-defined: detect, localize, or quantify findings in the image. Third, there are large, standardized datasets — PACS archives, public collections like NIH ChestX-ray14, and teaching files — that can be used for training.

The underlying computer-vision machinery is covered in our computer vision primer. In radiology, those generic techniques are specialized for medical imaging: handling DICOM format, 3D volumes (not just 2D images), grayscale intensities carrying clinical meaning, and clinical-context integration with the rest of the patient record.
The main deployed use cases
Triage and worklist prioritization
AI models scan images as they arrive and flag potential critical findings — intracranial hemorrhage on head CT, large vessel occlusion (LVO) stroke, pulmonary embolism, pneumothorax. The flag moves the case to the top of the radiologist’s worklist, reducing time-to-read for high-acuity cases from hours to minutes. Companies like Aidoc, Viz.ai, RapidAI, and Zebra Medical Vision dominate this category.
Detection assistance (“second reader”)
Mammography was the first domain to use AI as a second reader, dating back to older feature-engineered systems in the 2000s and now deep-learning-based. The model reviews the image and flags suspicious areas for the radiologist’s attention. Chest X-ray, lung CT (for nodule detection), and colonoscopy (for polyp detection during the procedure) have similar tools.
Quantification
Precise measurement is harder for humans than binary detection. AI quantifies tumor volumes across follow-up scans, measures bone mineral density from CT, estimates left-ventricular ejection fraction from echocardiography, and tracks brain atrophy in multiple sclerosis. These metrics support treatment decisions that would be difficult to make from visual impression alone.
Protocoling and scanner optimization
AI can accelerate MRI acquisition by reconstructing high-quality images from undersampled data — reducing scan time from 30+ minutes to under 10 for some sequences. GE, Siemens, and Philips all ship AI-powered reconstruction now.
How the models are trained
A typical radiology AI starts with a large labelled dataset — often hundreds of thousands of scans with expert annotations. Annotation is expensive; labelling a chest X-ray with pixel-level lung boundaries takes a radiologist minutes per image. Modern pipelines use semi-supervised techniques, self-supervised pre-training, and weakly supervised learning from radiology reports to reduce the labelling burden.
The model architecture is typically a convolutional backbone — U-Net for segmentation, ResNet or EfficientNet for classification — increasingly augmented with or replaced by vision transformers. The outputs are class probabilities, bounding boxes, or pixel-level segmentation masks. See our deep learning explainer for the underlying techniques.
Validation and regulatory path
In the US, radiology AI devices typically go through FDA’s 510(k) pathway — demonstrating substantial equivalence to a predicate device — or, for truly novel capabilities, the De Novo pathway. Clinical validation requires retrospective testing on held-out patient cohorts and, increasingly, prospective multi-site studies. The American College of Radiology’s Data Science Institute has pushed for more rigorous real-world performance evaluation.
The European Union’s MDR (Medical Device Regulation) imposes comparable requirements. Both systems are wrestling with how to handle models that update over time — so-called “continuously learning” systems — though in practice most deployed models are locked at clearance and update only with new submissions.
Does AI outperform radiologists?
Benchmark studies have shown AI matching or exceeding radiologist performance in narrow tasks — diabetic retinopathy screening, single-lesion mammography detection, pneumothorax detection on chest X-ray. But benchmark performance is not clinical performance. Real-world studies have been more nuanced: AI can match average radiologists on average cases but often underperforms on unusual presentations, edge cases, and outside the training-data distribution.
The radiology community’s consensus has shifted from the 2016-era “replacement” framing to collaboration. AI catches routine findings quickly; the radiologist focuses attention on complex cases, synthesizes multi-modal information, and bears clinical judgment responsibility. Jobs have not disappeared — the American College of Radiology projects ongoing radiologist shortages. For a broader healthcare view, see our ai in healthcare coverage.
Ongoing challenges
Generalizability
A model trained at one academic medical centre may fail at a community hospital using different scanners, different patient populations, and different protocols. This “external validity” problem has led some sites to conduct mandatory local validation before deployment.
Reimbursement
Payers have been slow to reimburse for AI-augmented imaging. Without a reimbursement code, hospitals have to absorb the AI tool’s cost or justify it through efficiency gains. CMS (US Medicare) has added a small number of AI-related codes but coverage remains patchy.
Workflow friction
AI that requires opening a separate tool adds clicks. The winning products integrate directly into existing PACS and reporting systems so findings appear in the radiologist’s normal view.
Skepticism and alert fatigue
Radiologists who see many false-positive flags stop trusting the tool. Calibration — making sure confidence scores mean what they say — is as important as raw accuracy.
Emerging directions
Foundation models trained on large collections of medical images — like Google’s Med-Gemini, or research efforts in open-source medical vision models — are moving toward general-purpose radiology AI. Instead of one model per finding, one model handles many tasks and can be specialized with small fine-tunes. Multimodal models that combine images with clinical text (prior reports, lab values, patient history) are another active direction. The next decade will likely see AI-radiology tools go from point solutions to integrated platforms.
Frequently asked questions
Will AI replace radiologists?
Widely cited 2016 predictions that AI would eliminate radiology jobs within 5-10 years have not materialized. If anything, radiologist demand has grown, driven by rising imaging volume. AI is absorbing specific tasks — triage, quantification, some detection — but clinical decision-making, communication with referring physicians, interventional procedures, and judgment across modalities remain human work. The realistic near-term scenario is radiologists using AI tools to work faster and catch more, not being replaced by them.
Are FDA-cleared AI radiology tools proven to improve outcomes?
Clearance requires demonstration of safety and accuracy, not outcomes improvement. Few tools have published large-scale studies of actual patient outcomes. Triage tools for stroke and pulmonary embolism have the best outcomes evidence so far — faster treatment times demonstrably reduce morbidity. For most tools, the clinical-outcomes evidence is thin, which is a frequent topic in radiology journals and professional societies.
Can AI read an X-ray without a radiologist present?
For a few narrow applications, yes. IDx-DR screens for diabetic retinopathy autonomously and refers positive cases to an ophthalmologist. Most radiology AI, though, produces findings that a radiologist interprets in context. Fully autonomous reading is restricted to low-risk binary-outcome screening; diagnostic reading stays with a licensed physician.