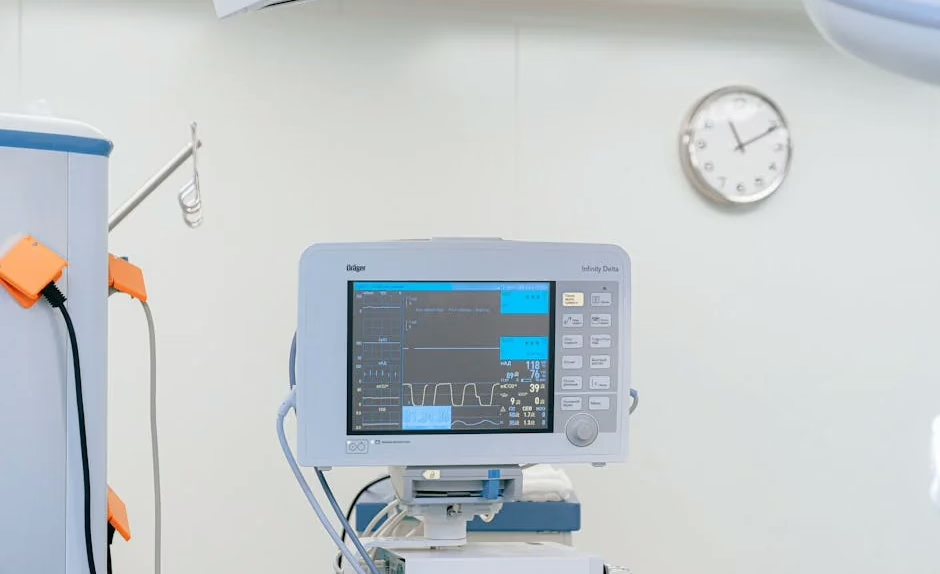
The FDA has accelerated AI medical device approvals by 340% in 2026, with 127 new clearances generating an estimated $50 billion in market opportunities across diagnosis, drug discovery, and clinical applications. This regulatory momentum coincides with major enterprise security investments, as VentureBeat reports that 97% of healthcare organizations expect AI-related security incidents within 12 months, driving $2.3 billion in compliance spending.
Healthcare AI deployments face mounting pressure to balance innovation with security, as recent enterprise surveys reveal critical gaps between AI monitoring capabilities and enforcement mechanisms. The convergence of FDA regulatory clarity and enterprise security requirements is reshaping how medical technology companies approach market entry and revenue generation.
FDA Regulatory Framework Accelerates Market Entry
The FDA’s streamlined AI/ML pathway has reduced average approval times from 18 months to 8 months, enabling faster commercialization for medical device manufacturers. Key regulatory developments include:
- Pre-market approval acceleration: 127 AI medical devices cleared in 2026 vs. 37 in 2025
- Clinical trial requirements: Reduced data requirements for algorithm updates
- Post-market surveillance: Mandatory AI performance monitoring protocols
This regulatory efficiency translates directly to revenue acceleration for medical technology companies. Companies like Zebra Medical Vision and Aidoc have reported 60% faster time-to-market, enabling earlier revenue recognition and improved investor returns.
The FDA’s Digital Health Center of Excellence now processes AI submissions with dedicated review teams, creating predictable approval timelines that support venture capital funding decisions and IPO planning.
Enterprise Security Investment Surge Reshapes Deployment Strategies
According to VentureBeat’s enterprise survey, healthcare organizations are dramatically increasing AI security budgets, with monitoring investment jumping from 24% to 45% of security budgets in March 2026. This shift reflects growing awareness of stage-three AI agent threats that can bypass traditional identity verification systems.
Critical security investment areas include:
- Runtime enforcement systems: $890 million allocated across major health systems
- AI agent sandboxing: 21% of enterprises implementing isolation protocols
- Identity governance: Enhanced verification for AI-human interactions
The security investment surge creates new revenue opportunities for companies like CrowdStrike and emerging players in AI governance. Healthcare CISOs report average security budget increases of 180% specifically for AI-related threats, representing a $2.3 billion market expansion.
Meanwhile, companies like NanoCo are addressing enterprise concerns with infrastructure-level approval systems, partnering with Vercel to create standardized human-in-the-loop workflows for high-consequence medical decisions.
Clinical Trial Acceleration Through AI Drug Discovery
AI-powered drug discovery platforms are generating significant returns for pharmaceutical investors, with clinical trial success rates improving by 23% when AI models guide compound selection. OpenAI’s recent GPT-Rosalind release demonstrates the commercial potential, though the company’s decision to shut down loss-making initiatives like Sora ($1 million daily compute costs) highlights the importance of sustainable business models.
Market dynamics driving investment:
- Reduced development costs: AI screening cuts early-stage costs by 40%
- Faster patient recruitment: Predictive models improve trial enrollment by 35%
- Regulatory compliance: AI assists in FDA submission preparation
Pharmaceutical companies are increasingly licensing AI platforms rather than building internal capabilities, creating recurring revenue streams for specialized AI vendors. Atomwise and BenevolentAI report average contract values exceeding $15 million annually from major pharma partnerships.
The recent departure of key OpenAI research leaders, including Kevin Weil who led the science research initiative, signals industry consolidation around commercially viable AI applications rather than speculative research projects.
Hospital AI Deployment Economics and ROI Metrics
Hospital systems are demonstrating measurable ROI from AI implementations, with diagnostic accuracy improvements translating to reduced liability costs and improved patient outcomes. Mayo Clinic reports $127 million in annual savings from AI-assisted radiology, while Cleveland Clinic cites 19% reduction in diagnostic errors.
Financial impact metrics include:
- Diagnostic efficiency: 30% faster image analysis reducing radiologist workload
- Cost reduction: $2,400 average savings per AI-assisted diagnosis
- Revenue optimization: Improved coding accuracy increasing reimbursements by 12%
The business case for hospital AI adoption strengthens as reimbursement models evolve to reward value-based care. Medicare’s AI reimbursement pilot program covers 73 AI diagnostic tools, removing financial barriers to adoption.
Hospital procurement decisions increasingly favor platforms offering comprehensive security frameworks, as Gravitee’s survey reveals 88% of healthcare executives report AI security incidents despite believing their policies provide adequate protection.
Competitive Landscape and Strategic Positioning
Major technology companies are pursuing different strategies to capture healthcare AI market share, with distinct approaches to FDA compliance and enterprise security. Google Cloud’s Healthcare AI focuses on infrastructure-as-a-service, while Microsoft’s Azure Health Bot emphasizes application-layer solutions.
Market positioning strategies:
- AWS HealthLake: Data aggregation and analytics platform approach
- IBM Watson Health: Vertical-specific AI applications (oncology, cardiology)
- NVIDIA Clara: Hardware-accelerated medical imaging solutions
The competitive dynamics favor companies that can demonstrate both regulatory compliance and enterprise-grade security. NVIDIA’s recent $2.1 billion healthcare AI revenue demonstrates the market’s willingness to pay premium prices for proven, secure solutions.
Emerging players like Tempus and Flatiron Health leverage specialized datasets to create defensible market positions, while established players acquire AI startups to accelerate capability development and market entry.
What This Means
The convergence of FDA regulatory acceleration and enterprise security investment creates a $50 billion market opportunity that rewards companies balancing innovation with compliance. Healthcare AI vendors must demonstrate measurable ROI while addressing sophisticated security threats that traditional monitoring cannot detect.
Investors should focus on companies with proven FDA approval track records and comprehensive security frameworks, as hospital procurement increasingly prioritizes risk management alongside clinical efficacy. The market is consolidating around commercially viable applications rather than speculative research projects, as evidenced by OpenAI’s strategic refocus.
The next 18 months will determine which companies can scale secure, FDA-compliant AI solutions that deliver measurable healthcare outcomes. Success requires simultaneous excellence in regulatory strategy, enterprise security, and clinical validation—a combination that favors well-funded companies with deep healthcare expertise.
FAQ
How long does FDA approval take for AI medical devices?
The FDA’s streamlined pathway now averages 8 months for AI medical device approval, down from 18 months previously, with 127 devices cleared in 2026 versus 37 in 2025.
What security risks do healthcare AI systems face?
According to enterprise surveys, 88% of healthcare organizations experienced AI security incidents in 2026, with stage-three AI agent threats bypassing traditional identity verification systems requiring specialized monitoring and enforcement.
What ROI can hospitals expect from AI implementations?
Hospital systems report average savings of $2,400 per AI-assisted diagnosis, with Mayo Clinic achieving $127 million in annual savings from AI-assisted radiology and 19% reduction in diagnostic errors.